Adult patients with advanced midline gliomas have been treated with a peptide vaccine for the first time, according to researchers from Heidelberg and Mannheim, Germany. The vaccine mimicked a mutational change in a histone protein (H3K27M) that is a marker for this difficult-to-treat type of brain tumor. The vaccine proved to be safe and induced a promising immune response.
“We have shown previously that this genetic alteration can be recognized by the immune system, specifically T helper cells, and that a vaccine results in the activation of specific T helper cells and subsequent control of tumors carrying this genetic alteration in mice,” co-senior author Michael Platten tells Inside Precision Medicine. Platten is director of the Department of Neurology at the University Medical Center Mannheim and Head of Department at the German Cancer Research Center (DKFZ).
The team reported their work in Nature Medicine. The first author is Niklas Grassi.
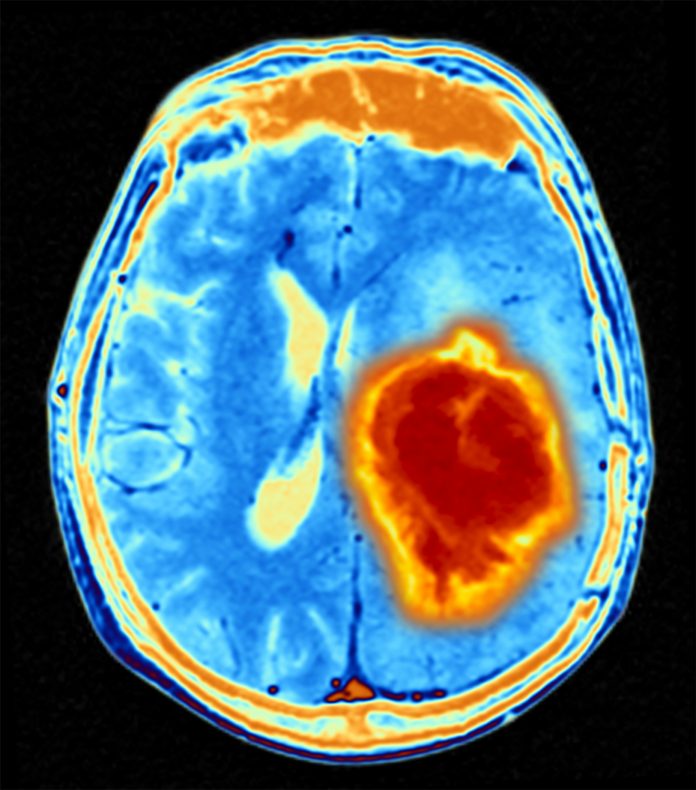
Diffuse midline gliomas are among the most aggressive brain tumors, with a life expectancy of less than one year. They usually occur in children and young adults near the brain stem and are difficult to access surgically. Chemotherapy or radiation therapy also have limited effectiveness.
Mutations in this type of tumor typically occur in H3K27M, which is a DNA packaging protein. The mutations produce a novel protein structure—a neoepitope, which the patient’s immune system can recognize as foreign. Substitution of lysine 27 to methionine in H3K27M is one mutation known to cause an aggressive subtype of diffuse glioma.
“Such mutations, which occur in identical form in many patients, are rare in cancer. They literally lend themselves to the development of tumor vaccines because they occur in all cancer cells, since the mutated histone is causative for the development of midline gliomas. This means that vaccination against the mutated protein gets to the root of the problem,” explains Platten.
The team, which was led by Platten and Katharina Sahm, senior physician at the Neurological University Hospital Mannheim and DKFZ researcher, synthetically reconstructed the section of the histone H3 protein with the characteristic mutation. Using this peptide, they curbed the growth of H3K27M-mutated tumors in a mouse model. They then tested the mutation-specific vaccine, produced at the University of Tübingen, in patients in a Phase I trial, which is still ongoing.
In parallel, the team treated eight adult patients with the peptide vaccine on a compassionate use basis. These patients had diffuse midline gliomas, with H3K27M mutation, that had progressed after standard therapy. Some of the affected individuals received therapy with immune checkpoint inhibitors in addition to tumor vaccination.
No serious side effects were observed in any of the vaccinated patients. Five of the eight developed specific immune responses against the mutant protein. These responses were dominated by CD4 T-helper cells. In one patient, who showed a strong immune response, the tumor regressed completely and she remained tumor-free for 31 months.
HLA proteins are responsible for the presentation of the mutant peptide on the cell surface and differ from person to person depending on the patient’s genetic background. The researchers report that this vaccine peptide, which is comparatively long at 27 amino acids, worked in patients with different HLA variants.
“…the current study has given us valuable information that will help us to further optimize the development of brain tumor vaccines in the future,” explains Sahm.
A Phase I trial is currently underway to test the vaccine against the H3K27M mutation in patients with newly diagnosed midline gliomas. Evaluation is expected to begin around 2025.