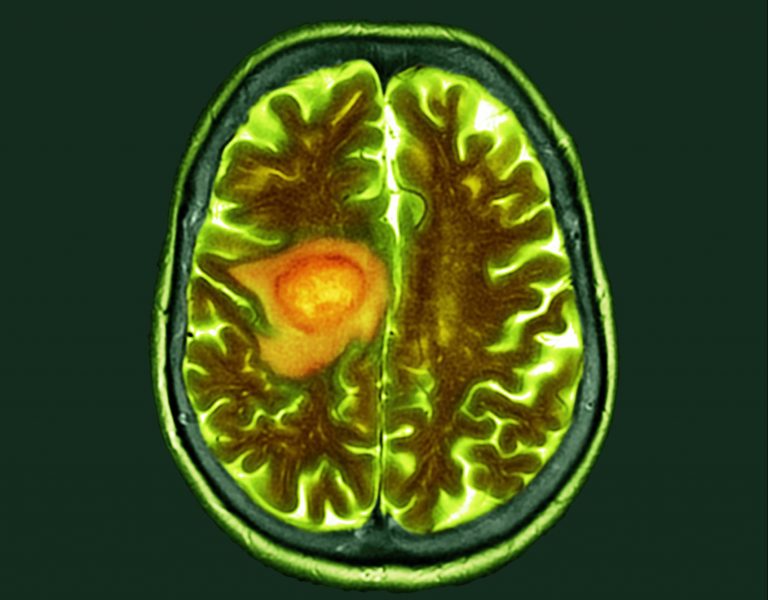
Researchers at the Medical College of Wisconsin Cancer Center have begun a clinical trial of a new, oral, gallium-based treatment for glioblastoma, an aggressive form of brain cancer.
The new trial is based on earlier pre-clinical studies led by Medical College of Wisconsin oncologist and professor Christopher Chitambar, which showed that gallium could bind to glioblastoma cancer cells and disrupt iron metabolism leading to their death.
Increased iron levels in the body can increase the risk for cancer and also increase chances for progression as iron is needed for cancer cells to proliferate. Chitambar and his colleagues have spent years researching iron metabolism in cancer cells and developing the new treatment, an orally available form of gallium called gallium maltolate (GaM).
In the body, GaM shares many properties with a highly oxidized version of iron, Fe (III). This allows it to enter cancer cells in the place of Fe (III) and destroy them instead of promoting their growth.
While GaM has fairly wide anti-cancer properties, the opportunity to treat glioblastoma is important due to the aggressive and resistant nature of this kind of tumor. “The discovery that GaM has anticancer activity against glioblastoma in pre-clinical studies is extremely exciting; it opens the door for developing it as a drug for treatment of glioblastoma in patients,” said Chitambar, in a press statement.
The phase I trial will initially treat 24 patients with relapsed glioblastoma with different doses of oral GaM to test anti-cancer activity, safety and tolerance of the drug.
Glioblastoma is an aggressive brain cancer, representing 15% of all brain cancers, and is hard to treat. Less than 7% of patients survive longer than five years and even if treatment is initially successful, most cases recur. If GaM is effective, it will help meet an unmet treatment need.
A number of other potential glioblastoma treatments are being developed, although most are in the early stages. For example, targeting and suppressing the protein galectin 1, activating DNA Fragmentation Factor 40, or the production of specially targeted CAR T-cell therapy, among others.
Chitambar and his colleague Jennifer Connelly are both principal investigators on the trial, which is being supported by the biotech and imaging firm Imaging Biometrics, as well as the Musella Brain Tumor Foundation and the MCW Cancer Center. Imaging Biometrics will provide the imaging and imaging analysis needed for the trial, which has a planned completion date of December 2025.