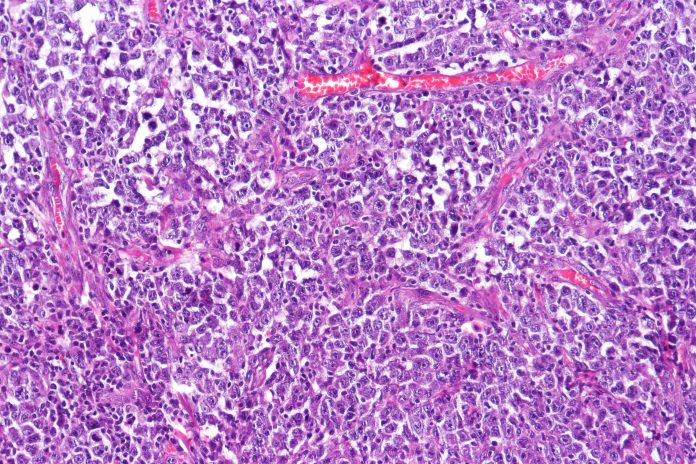
Researchers from Weill Cornell Medicine and New York-Presbyterian have identified a surprising mechanism that ultimately allows for DNA repair in cancer cells and in their both fundamental research and clinical studies have found a combination of chemotherapies that address these treatment-resistant lymphomas. Details of this approach, which is published in the journal Cancer Research, centered on diffuse large B-cell lymphoma (DLBCL), but is likely applicable to other cancers as well.
DLBCL affects roughly 30,000 every year in the U.S. and is the most common form of the disease. First line therapies cure about two-thirds of all patients “but for people who aren’t cured or who relapse, they historically had poor outcomes with standard chemotherapy-based treatment approaches,” said co-lead author Sarah Rutherford, MD, an assistant professor of medicine at Weill Cornell Medicine and a hematologist/oncologist at NewYork-Presbyterian/Weill Cornell Medical Center.
A mechanism discovered in the research involves the shuttling of messenger RNAs (mRNAs) from the cell nucleus to the cytoplasm where it can then facilitate the repairs of DNA in cancer cells allowing these cells to resist treatments that are designed to fight the cancer via damaging the cells’ DNA.
Previous research has shown that treatment-resistant DLBCL often express high levels of the protein XPO1. This protein is targeted by the drug Selinexor—approved by the FDA in 2019—which inhibits the protein’s activity and, as a result, the growth of the lymphoma cells expressing high levels of XPO1. Selinexor has helped many, but not all patients with treatment resistant DLBCL.
As Rutherford noted “Selinexor is effective by itself, it’s just not as effective as we’d like it to be,” which has prompted additional research to find ways to make the treatment effective for more patients.
Research aimed at better understanding exactly how selixinor works by Rutherford’s colleagues revealed that XPO1 serves as a method of transporting hundreds of proteins out of the nucleus functioning to separate those proteins that should not be present in the cell nucleus such as ribosomal proteins. What these researchers also found was that some of the proteins shepherded out of the nucleus by XPO1 also bound to mRNA. The effect of this is the mRNAs are exported into the cytoplasm where they can be translated into protein, with the quantity and activity of XPO1 thereby affecting the expression level of numerous genes within the cell.
“We found that it’s not just regulating a few proteins, it’s coordinating these big programs, allowing cells to rapidly adjust their proteome and survive different kinds of stress that cancer cells encounter all the time,” said senior author Leandro Cerchietti, MD, an associate professor of Hematology and Oncology and a member of the Meyer Cancer Center at Weill Cornell Medicine.
Building on this knowledge, the team then took sample of treatment-resistant DLBCL cells from patients and grafted unto preclinical models. This that revealed higher levels of XPO1 lead to processes that eventually increase the expression of genes that protect cancer from DNA damage that leads to cell death. Inhibiting XPO1 in those models with Selinexor increased the lymphomas’ sensitivity to DNA-damaging chemotherapies and immune-based treatments.
“Based on some of Cerchietti’s research, we thought Selinexor would likely synergize with other chemotherapies,” said Rutherford. To test that in patients, she initiated a Phase I clinical trial, aiming to determine whether a combination therapy approach would be safe, and if so at what doses. The trial, which primarily enrolled patients with treatment-resistant DLBCL, showed that the combined regimen is not only safe, but appears to work. The team will now conduct follow up trials, since the data set from the initial study was too small from which to draw definitive conclusions.
Further, this continued research could have implications for other treatment-resistant cancers. “There are other tumors in which XPO1 is overexpressed, so it’s really a nice backbone to build on,” said Cerchietti.