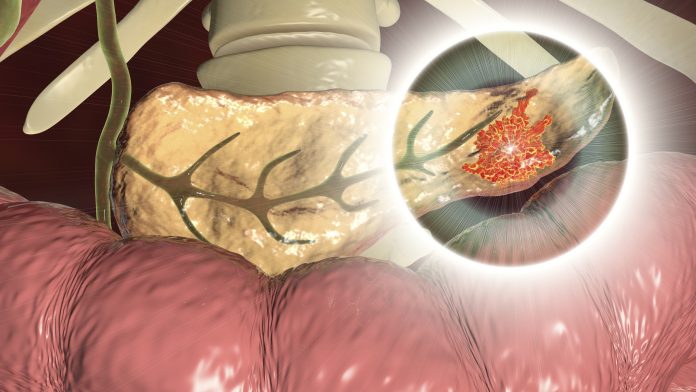
Researchers from Harvard Medical School (HMS) and the University of Copenhagen, in collaboration with VA Boston Healthcare System, Dana-Farber Cancer Institute, and the Harvard T.H. Chan School of Public Health say they have developed an AI model that predicts pancreatic cancer risk three years before diagnosis. The report detailing the new model, which analyzes only a patient’s medical record, was published this week in Nature Medicine.
The new model could be used as a screening tool to hasten pancreatic cancer diagnoses—a cancer known for its high mortality since it is usually discovered when it is at a late stage—and improve treatments by starting them earlier. About 44% of people diagnosed in the early stages of pancreatic cancer survive five years after diagnosis, but only 12% of cases are diagnosed that early. The survival rate drops to 2% to 9% for those whose tumors have grown beyond their site of origin, researchers estimate.
“One of the most important decisions clinicians face day to day is who is at high risk for a disease, and who would benefit from further testing, which can also mean more invasive and more expensive procedures that carry their own risks,” said study co-senior investigator Chris Sander, faculty member in the Department of Systems Biology in the Blavatnik Institute at HMS. “An AI tool that can zero in on those at highest risk for pancreatic cancer who stand to benefit most from further tests could go a long way toward improving clinical decision-making.”
The tool could be the first population-based screening method for pancreatic cancer and pick up those that fall outside the current standard of targeted screenings based on family history, or a specific genetic profile.
“Many types of cancer, especially those hard to identify and treat early, exert a disproportionate toll on patients, families and the healthcare system as a whole,” said study co-senior investigator Søren Brunak, professor of disease systems biology and director of research at the Novo Nordisk Foundation Center for Protein Research at the University of Copenhagen, in a press release. “AI-based screening is an opportunity to alter the trajectory of pancreatic cancer, an aggressive disease that is notoriously hard to diagnose early and treat promptly when the chances for success are highest.”
Model training
To train the AI algorithm, researchers used two data sets with a total of 9 million patient records from Denmark and the United States. The AI model was built to look for telltale signs based on the data in the records, on combinations of disease codes, and the timing of their occurrence, to predict the patients most likely to develop pancreatic cancer in the future. Notably, many of the symptoms and disease codes were not directly related to or stemming from the pancreas.
The researchers tested different versions of the AI models for their ability to detect people at elevated risk for disease development within different time scales—6 months, one year, two years, and three years. Overall, each version of the AI algorithm was substantially more accurate at predicting who would develop pancreatic cancer than current population-wide estimates of disease incidence—defined as how often a condition develops in a population over a specific period of time. The researchers believe the model is at least as accurate in predicting disease occurrence as are current genetic sequencing tests that are usually available only for a small subset of patients.
For the current study, the researchers designed several versions of the AI model and trained them on the health records of 6.2 million patients from Denmark’s national health system spanning 41 years. Of those patients, 23,985 developed pancreatic cancer. During the training, the algorithm discerned patterns indicative of future pancreatic cancer risk based whether the patient had certain conditions that occurred in a certain sequence over time.
For example, diagnoses such as gallstones, anemia, type 2 diabetes, and other GI-related problems portended greater risk of developoing pancreatic cancer within 3 years of evaluation. Inflammation of the pancreas was strongly predictive of future pancreatic cancer within an even shorter time span of two years. The researchers caution that none of these diagnoses by themselves should be deemed indicative or causative of future pancreatic cancer. However, the pattern and sequence in which they occur over time offer clues for an AI-based surveillance model and could prompt physicians to monitor or test those at elevated risk.
The researchers then sought to confirm the AI model’s predictive capabilities, by using the best performing model on a new set of patient records obtained from a U.S. Veterans Health Administration data set of nearly 3 million records spanning 21 years and containing 3,864 individuals diagnosed with pancreatic cancer. While the model’s accuracy was a bit lower with these records, the researchers suggest it was due to the shorter time span of records available for analysis.
Detecting pancreatic cancer earlier
Pancreatic cancer is harder and more expensive to screen and test for compared with other cancers which have effective screening methods available. Typically, for pancreatic cancer, doctors will use information on a patient’s family history and/or the presence of specific genetic mutations which have been linked to the disease. The advantage of an AI tool is that it could be used on any patient by simply examining their health records. This is especially important, the researchers add, because many patients at high risk may not even be aware of their genetic predisposition or family history.
In the absence of symptoms and without a clear indication that someone is at high risk for pancreatic cancer, clinicians may be understandably cautious to recommend more sophisticated and more expensive testing, such as CT scans, MRI or endoscopic ultrasound. When these tests are used and suspicious lesions discovered, the patient must undergo a procedure to obtain a biopsy. Positioned deep inside the abdomen, the organ is hard to access and easy to provoke and inflame.
An AI tool that identifies those at the highest risk for pancreatic cancer would ensure that clinicians test the right population, while sparing others unnecessary testing and additional procedures, the researchers said.
“That low survival rate is despite marked advances in surgical techniques, chemotherapy, and immunotherapy,” Sander said. “So, in addition to sophisticated treatments, there is a clear need for better screening, more targeted testing, and earlier diagnosis, and this where the AI-based approach comes in as the first critical step in this continuum.”