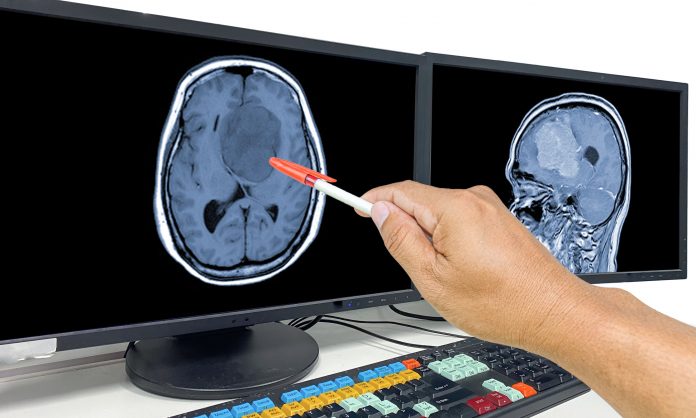
Researchers achieved promising results treating glioblastoma (GBM) in an innovative clinical trial. The trial utilized a modified oncolytic adenovirus combined with immunotherapy to target and destroy cancer cells via apoptosis while stimulating the immune system to kill residual cancer cells. The Phase I/II study, conducted by neurosurgeons at the University Health Network in Toronto, showed significant improvements in patient survival rates and tumor shrinkage. The results of the trial are published in Nature Medicine.
The team evaluated the combination of intratumoral delivery of oncolytic virus DNX-2401 followed by intravenous anti-PD-1 antibody (pembrolizumab) in 49 patients with recurrent glioblastoma, first in a dose-escalation regimen, and then in a dose-expansion phase.
The modified adenovirus, developed by Dr. Juan Fueyo and Dr. Candelaria Gomez-Manzano of the University of Texas MD Anderson Cancer Center, is specifically designed to replicate within glioblastoma cells, sparing healthy brain cells from infection.
Immune checkpoint blockade in other advanced solid cancers such as melanoma and non-small cell lung cancer has greatly improved outcomes. However, the immunologically cold microenvironment in glioblastomas has presumably rendered immune checkpoint blockade less effective for this disease.
The combination of adenovirus and immunotherapy demonstrated overall survival of 52.7% at 12 months. Median overall survival was 12.5 months (10.7–13.5 months), compared to approximately three months for standard treatments. A total of 56.2% of patients had a clinical benefit defined as stable disease or better.
“Another exciting result is that in a subset of patients, we actually saw the tumors shrink substantially to 50% or more of the original size and none of these patients had any tumor resection,” said first author Farshad Nassiri, MD, senior neurosurgery resident at the University of Toronto. “And for two of the patients, the tumors went completely away.”
Three patients completed treatment with durable responses and remain alive at 45, 48, and 60 months. The treatment was well-tolerated with no dose-limiting toxicities.
Unlike traditional methods of injecting viruses into the tumor cavity following resection, the researchers injected the adenovirus directly into the tumor through a small hole and a purpose-built catheter. Patients received anti-PD-1 antibody every three weeks, starting one week after surgery.
“Along with inducing apoptosis in cancer cells, the virus causes an immune reaction within the microenvironment causing additional immune cells to flood in,” added Nassiri. “Then we give the systemic immunotherapy and essentially allow it to work. The virus turns the ‘cold’ tumor into an immunologically active tumor.”
Previous attempts at using immunotherapy alone have had limited efficacy due to the low presence of immune cells in the tumor microenvironment and the challenge of overcoming the blood-brain barrier.
“By directly injecting the virus into the tumor, we bypass the blood-brain issue altogether,” said senior author Gelareh Zadeh, MD, PhD, professor in the Department of Surgery at the University of Toronto.
The team emphasizes that they did not remove any tumor prior to injecting the virus. In the past, viruses injected into the tumor cavity failed to produce promising results. “If you think about how the virus works, it infects cancer,” adds Zadeh, “so if you remove the bulk of the cancer cells, there’s not very much for the virus to infect to trigger cancer cell death.”
The team also discovered a unique molecular signature that differentiated responders from non-responders in the trial. Patients with an ‘intermediate’ immune signature had the most favorable outcomes. These were the patients whose tumors were not immunologically cold or so immunologically active that the immunotherapy either had nothing to bind to or was overrun by anti-immune signaling that is also triggered in the presence of overly abundant immune cells.
“This indicates the importance of striking a balance between having some solid tumor there for the virus to be able to have an effect that can then be augmented by immunotherapy,” says Zadeh. “It is very exciting to be able to see a signature within a clinical trial that predicts results, but I think it’s even more exciting when the signature makes biological sense.”
This finding has opened the door to the potential for tailored therapies for individual patients. When the team studied tissues before and after treatment, they observed multiple different complementary immune checkpoints being activated in different patients. “For these patients, perhaps one can use different combinatorial therapies, different combinations of monoclonal antibodies, to block multiple different pathways,” noted Nassiri. “Potentially you can overcome the resistance that they have to single agent immune therapies, even when they’re augmented with the oncolytic virus.”