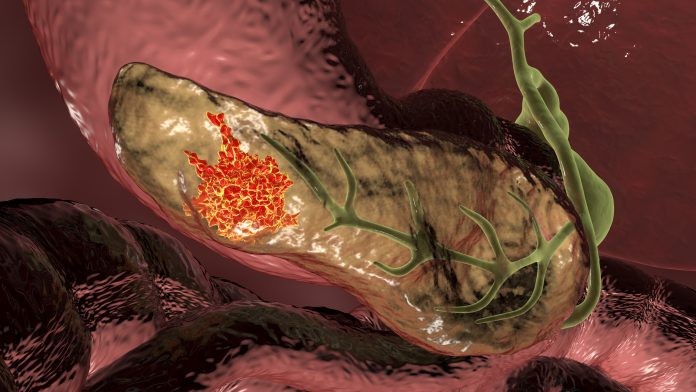
Combining radiation with a novel immunotherapy drug was shown to stop metastasis in animal models of pancreatic ductal adenocarcinoma (PDAC), which accounts for 90% of pancreatic cancers. According to a new study by researchers at the University of Colorado Cancer Center published in Cancer Cell, the therapy resulted in significantly improved survival. They hope this treatment will improve survival rates for a disease that has not historically responded to immunotherapy.
At the heart of the therapy is a novel variant antibody complex (aPD1-IL2v), developed by Roche and described in Nature. The antibody is engineered such that it will bind only to CD8 T cell effector cells—the main anticancer immune cells—but not the immunosuppressive T cells, or T regulatory cells (Tregs).
“One of the many problems with immunotherapy is how do you make it target just the effector T cells and avoid the immunosuppressive T cells,” said lead author Sana Karam, MD, PhD. This antibody appears to boost the expansion of CD8 T cells to fight cancer without triggering Treg activity, which she refers to as ‘next-generation’ immunotherapy.
The antibody takes advantage of the IL-2 signaling pathway, which is well known to have a key role in promoting the survival of T cells. IL-2 provides the fuel for both CD8 cells and Tregs. However, because the drug only binds to CD8 T cells, it deprives Tregs from using IL-2.
“The antibody gives the ability for the CD8 T cells to continue to use the IL-2 A while the Tregs are not able to,” said Karam. The Tregs remain but they are not functional any longer because they are deprived of their IL-2 fuel – they are not hijacking the IL-2 away from the CD8 cells.
The binding properties of the drug avoid regulatory T cells since it is specific only to CD8 T cells. For this reason, the antibody is known as an IL-2 variant antibody. “This is a specific IL-2-driven amplification for just CD8 effector cells and that’s where the brilliance of the design is,” added Karam. “So we see a massive out-of-this-world expansion of CD8 T cells whether it be in the tumor and more importantly, in the blood,” she adds.
This finding is important since the driver of morbidity and mortality in pancreatic cancer is metastasis. By this enormous expansion of memory effector T cells in the blood, it gives the ability to not only deal with a tumor locally, but also distantly in metastasis. The team found that the antibody can recognize and kill the cancer cells, whether they are in the tumor microenvironment, in circulation, or in the lymph nodes. “It crosses all three compartments to bind to and destroy cancer cells,” said Karam.
In the past, researchers have attempted to manipulate the IL-2 pathway upwards for cancer therapy. But the strategy results in vascular leak syndrome and cytokine storm because the Tregs go on a feeding frenzy and proliferate.
The team studied the antibody in combination with just one dose of radiation therapy. “The two together gave us this beautiful synergy,” said Karam. The radiation increases the number of dendritic cells, which are known to have an immunostimulatory effect in cancer. “We have shown that it jacks up the recruitment and activation of dendritic cells both in patients and preclinical models,” she added. The dose is one she considers barely palliative with little or no side effects.
Karam has also studied the combination using patient samples from her own clinic. When they analyzed tissue and blood samples before and after radiation and antibody treatment, the CD8 T cells were highly amplified. The combination treatment is now under study in early Phase I safety and dosing studies at other sites.
Similar immunotherapy research aimed at other cancers, including melanoma and neuroendocrine tumors, is being conducted in other laboratories.
“I am very excited about this treatment, particularly as a physician-scientist,” added Karam. “I will not sit still until I can get this into my patients, it is my immediate goal in life.”