Henry Ford Health researchers have announced the initiation of a Phase I clinical trial to determine maximum tolerated dose of an oncolytic adenovirus in combination with fractionated stereotactic radiosurgery for patients with recurrent high-grade astrocytoma undergoing resection.
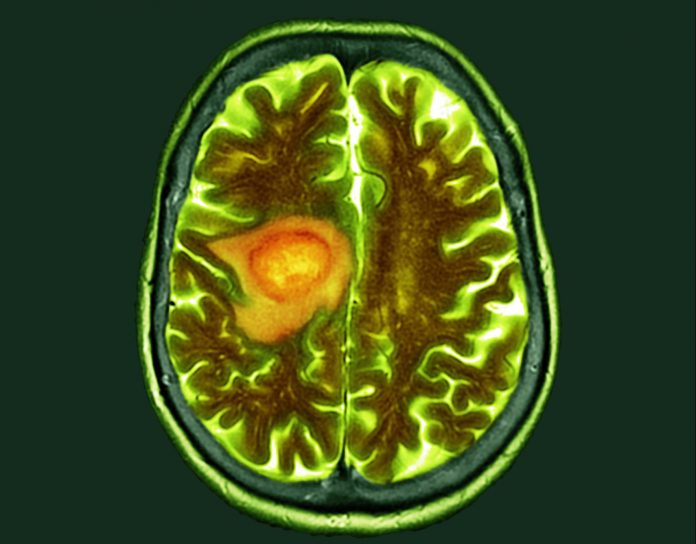
Astrocytomas are the most common type of primary brain tumors. Among them, the biggest and most aggressive is the group of grade 4 tumors known as glioblastomas.
“Despite best treatment, which generally is made up of best surgical resection, followed by radiation with chemotherapy and adjuvant chemotherapy, these tumors recur and so there’s an urgent need for new treatments,” Tobias Walbert, M.D., Ph.D., principal investigator of the study and co-director of the Hermelin Brain Tumor Center at Henry Ford Health told Inside Precision Medicine.
Henry Ford Health has over 25 years of development experience with oncolytic viruses. For the current study, they will use an in-house adenovirus type 5 “that has the ability to replicate within the cancer cell and cause lysis or the complete destruction of the cancer cell,” explained Farzan Siddiqui, M.D., Ph.D., co-investigator of the study and radiation oncologist at Henry Ford Health.
Along with its natural oncolytic properties, the virus, which has already been trialed in patients with metastatic pancreatic cancer and intermediate-risk prostate cancer, has been modified to include two “suicide genes”—yeast-cytosine deaminase and herpes simplex virus thymidine kinase.
When switched on, these genes convert the oral prodrugs 5-fluorocytosine and valganciclovir into the active chemotherapeutic agents 5-fluorouracil and ganciclovir monophosphate, respectively, in the tumor microenvironment.
“The term suicide gene refers to the fact that the cancer cells are turning on these genes within themselves when the oral prodrugs are taken, which allows conversion into the active forms of these drugs, that then kill the cells.” said Siddiqui. “The cells are essentially inviting trouble for themselves.”
The first few patients have already been enrolled in the trial, with plans for nine to 18 to be treated in total.
After the removal of as much tumor tissue as possible, the oncolytic adenovirus is injected directly into the wall of the resection cavity and any residual tumor tissue, allowing it to replicate in the local microenvironment so that it does not disseminate systemically and cause toxicity.
Along with the prodrug therapy, participants will also be treated with fractionated radiosurgery. Walbert said that using the adenovirus treatment in combination with radiosurgery is a unique aspect of the study that, based on preclinical data, he believes will enhance the efficacy of the virus by increasing activation and sensitizing the glioma cells.
The team are already planning further studies in patients with glioblastoma, including the development of a tracer for the virus so that they can see where it goes and what it does within the brain.
Siddiqui is hopeful that the treatment could ultimately gain regulatory approval. “Once we’ve shown the safety, then we can move on to the efficacy and even if we have a minimal signal of efficacy with this approach, there’s so little other promising therapies in this space that we feel that we will get the green light from the FDA going forward,” he remarked.