The U.S. Food and Drug Administration (FDA) has approved Vertex and CRISPR Therapeutics’ Casgevy (exa-cel), a CRISPR/Cas9 gene-edited cell therapy, for the treatment of transfusion-dependent beta thalassemia (TDT) in patients 12 years and older. This is the latest sign the gene and cell therapy market is maturing. This market is already estimated to be worth $15B and expected to grow to more than $80B over the next ten years or so.
Just a few weeks ago the FDA approved Casgevy to treat patients with sickle cell disease. Simultaneously, Bluebird Bio received the FDA OK for Lyfgenia, its gene therapy for sickle cell. In November, the U.K. Medicines and Healthcare products Regulatory Agency (MHRA) granted conditional marketing authorization for Casgevy in both sickle cell disease and TDT.
With this nod, Casgevy becomes the first treatment for TDT available in the U.S. using CRISPR gene-editing technology. TDT is an inherited blood disease that causes low levels of functioning hemoglobin, which carries oxygen.
“On the heels of the historic FDA approval of Casgevy for sickle cell disease, it is exciting to now secure approval for TDT well ahead of the PDUFA date,” said Reshma Kewalramani, MD, CEO and president of Vertex. “TDT patients deserve new, potentially curative treatment options.”
The cell and gene therapy pipeline is already pretty full, with thousands of trials ongoing. Several are already in Phase II or III, including treatments for cancers and amyloidosis.
Casgevy administration requires experience in stem cell transplantation. As a result, Vertex is establishing a network of independently operated, authorized treatment centers (ATCs) throughout the U.S. to offer the treatment. All nine current U.S. ATCs can offer Casgevy to eligible patients with either TDT or sickle cell disease. Additional ATCs will be activated in the coming weeks and a complete list of ATCs can be accessed at CASGEVY.com.
TDT requires frequent blood transfusions and iron chelation therapy throughout a person’s life. Due to anemia, patients may experience fatigue and shortness of breath, and infants or develop failure to thrive, jaundice, and feeding problems. Complications of TDT can also include an enlarged spleen, liver and/or heart, misshapen bones and delayed puberty.
Patients have reduced life expectancy, decreased quality of life, and lowered lifetime earnings and productivity. In the U.S., the median age of death for patients living with TDT is 37 years. Stem cell transplant from a matched donor can cure the condition, but this option, but is only available to a small fraction of people living with TDT because of the lack of available donors.
Vertex and CRISPR Tx charge $2.2 million for a one-time Casgevy treatment. The companies estimate that lifetime healthcare costs to manage TDT in the U.S. exceed $5 million.
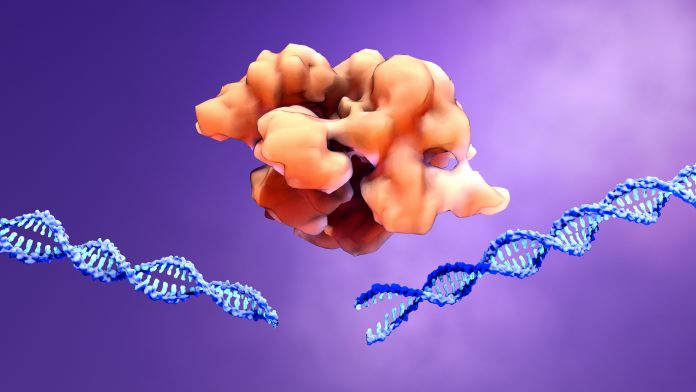
Casgevy is a non-viral, ex vivo CRISPR/Cas9 gene-edited cell therapy for certain patients with sickle cell disease or TDT. A patient’s own hematopoietic stem and progenitor cells are extracted and edited. The edits are made at the erythroid specific enhancer region of the BCL11A gene through a precise double-strand break.This change leads to production of high levels of fetal hemoglobin (HbF) in red blood cells. HbF is the form of hemoglobin typically present during fetal development, which then switches to the adult form after birth.